現在位置 : 醫療 > 心臟病 > 體外「心臟震波」治療冠心病 - CSWT (Cardiac Shock Wave Therapy)
Click here to edit.
|
體外「心臟震波」治療冠心病
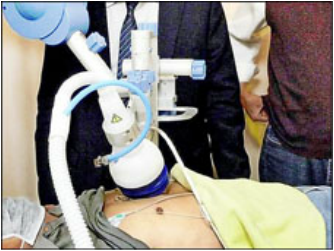
『心臟震波』 現生機 亞東醫院院訊 心臟血管內科 林恆旭醫師 近年台灣經濟發展快速,民眾生活逐漸傾向西方模式的飲食習慣與生活方式,使得心血管疾病比例日益增加。此外,隨著高齡化社會的來臨,伴隨年齡增長而來的身體各器官系統(尤其是心血管系統)的老化問題接踵而至,造成的醫療照護支出及社會成本逐年快速增加,目前心血管相關疾病已成為我國十大死因及健保就診人數之大宗,付出之醫療及社會相關成本也日益水漲船高。 一般冠心病(即心血管阻塞)病人常見的治療方式除了藥物治療外,還有心臟內科的心導管手術,利用氣球擴張或置放金屬支架來打通阻塞的血管,或是較為嚴重的患者可以選擇接受心臟外科冠狀動脈繞道手術,病患可依不同的狀況,選擇合適的治療方式。儘管上述治療經過數十年的發展,無論是經驗或技術上都已經相當成熟,但目前仍有約百分之十的冠心病病患(特別是合併有糖尿病患者),因冠狀動脈已產生瀰漫性病變,甚至部分病人的身體情況不佳,害怕手術帶來的相關併發症,並不完全適宜接受外科繞道手術或心導管手術治療,只能靠僅有的藥物來減緩心絞痛的發生,這些患者是目前冠心病族群中最缺乏醫療資源,也是治療上最-棘手的一群,這類病患明白藥物已不能完全解決自己的心臟問題,所以每天活在高度警戒的環境中,生活品質也因此大受影響。 「心血管爛爛的,不能縫不能補,心臟跳到都快喘不過氣來了,怎麼辦? 」 「當醫生宣布不能再動心導管和繞道手術時,心情馬上降到谷底!」 黃先生近年來因反覆胸悶至心臟科門診求診,經心導管確診為嚴重冠狀動脈阻塞,前後接受過三次心導管手術均無法順利打通病灶,考慮到繞道手術的風險又相當畏懼,本以為未來只能靠藥物治療。因緣際會之下,輾轉轉介至本院接受心臟體外震波治療,沒想到症狀於治療後一個月左右開始出現改善,在完成九週療程及藥物持續治療下,除了症狀明顯改善外,後續追蹤的核子醫學心臟灌注掃描以及心電圖均顯示心肌缺氧以及心律不整有顯著進步,大大減輕了冠心病為生活帶來的不安。 體外心臟震波治療是透過一種高頻率的聲波,和用於治療泌尿道結石的震波類似,但能量約只有泌尿科使用的1/10、促進骨癒合震波的1/4,也因為臨床上發現較低能量的震波可以增進骨頭或傷口癒合,才轉而朝治療冠心病發展。透過體外震波的刺激,如「體外按摩心臟」般,經由心臟超音波指引,以每平方毫米0.09毫焦耳的震波能量釋出,讓心臟產生新生的細小血管,進而改善心肌血流的灌注,減少心絞痛或心臟衰竭的發生。「心臟可比擬做水田,主渠道不通,讓小渠道去灌溉,一樣可以有足夠的血流量」。目前體外心臟震波初步治療對象鎖定為無法接受開刀或被醫師宣判無法動手術的患者,每次完整療程約1個小時,1療程每星期3次,每1個月1療程,共進行9次。因整體治療過程是透過心臟超音波定位,採體外震波方式,所以毋須開刀,也不用住院,只要定期回診即可。對於無法開刀或懼怕心導管及手術的患者來說是一大福音。台灣方面,也在去年通過衛福部核可後正式引進,唯目前健保局尚未核准給付這種新的治療方式,所以必須自費接受治療。 雖然心臟震波治療方式比起傳統心導管或心臟繞道手術更為簡便,但也不是每一個人都適合這種治療方式。如患者有心臟內血栓或急性心內膜炎、心肌炎等等,都不適合心臟震波的治療。必須經過心臟專科醫師為患者完整檢查並評估狀況後,方能決定採用何種治療方式。此外,患者必須認知,不論接受何種治療,治療後如不節制飲食、不運動,或者繼續抽菸,血管再度狹窄的可能性還是很高。遵從醫師指示、定時服藥、定期回診、定期量血壓及追蹤血糖、膽固醇,並配合生活作息調整來減少冠心病危險因子,才能讓治療的效果更顯著,也才是維護心臟健康的根本之道。 體外心臟震波治療情況 心臟體外震波治療成果之發表記者會現場 |
|
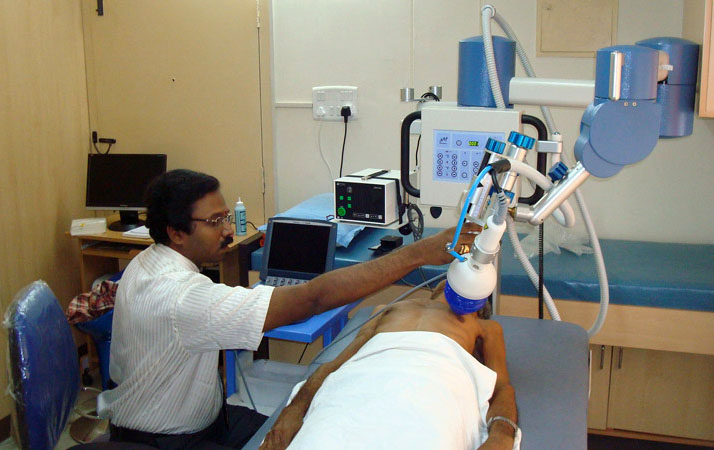
心絞痛新治療!心臟震波免開刀 華人健康網 記者張世傑/台北報導 2014年1月22日 冠心病是冬季健康殺手,特別是無預警的發作,更易導致高風險死亡 … 冠心病是健康隱形殺手,特別是有部分病況較嚴重患者,除了藥物外,因年紀、血管脆弱等無法進行手術治療,面臨死亡高風險威脅,目前亞東醫院引進「心臟震波」新治療,利用高頻率聲波產生小血管,不僅免開刀住院,更能減少心絞痛或是心臟衰竭的發生風險。(影音/攝影記者張世傑) 亞東醫院心臟血管內科主任吳彥雯醫師表示,近10年來,國人飲食習慣改變,導致罹患心血管相關疾病的人數增加,根據國民健康署公佈的國人10大疾病死亡原因調查,因心臟相關疾病死亡的人數蟬聯居整體排名第2,其中冠狀動脈心臟病(又稱為缺血性心臟病,簡稱冠心病),為心臟疾病死因中的主要原因。 心臟震波 緩心肌缺氧症狀 日前1名65歲黃姓男子,因反覆胸悶求診,經心導管確診為嚴重冠狀動脈阻塞,雖曾接受3次心導管手術仍難改善病灶,且血管狀況不佳,醫師推測就算接受冠狀動脈繞道手術也無法改善,後轉介至亞東醫院接受心臟體外震波治療。 臨床治療結果顯示,患者原本有大片心肌缺氧與心律不整,經過9周療程與藥物治療,症狀明顯改善,後續追蹤心臟灌注掃描與心電圖,影像顯示心肌缺氧與心律不整皆有明顯進步;而患者接受震波治療後,從原先稍微走路就覺得喘、心臟悶痛不適,到治療結束後可快走操場3至4圈,超音波檢查顯示心臟功能改善,核子醫學心肌灌流掃描也有進步。 高頻率聲波 能改善心肌血流 吳彥雯醫師強調,心臟好比水田,需要一定水量灌溉,但當主要灌溉渠道阻塞無法疏通時,可考慮挖通一些細小渠道,就如震波刺激心臟長出新生血管,幫助心臟提升血流灌溉量,改善心肌缺氧。 心臟震波治療無需麻醉、住院,但患有慢性肺部疾病、急性心內膜炎,或是其他全身性疾病與孕婦皆不適合接受治療。雖然心臟震波,可改善心肌血流灌流,但患者仍須遵從醫師指示,搭配其他治療方式,如藥物治療、飲食控制與生活作息調整等,才能提升治療效果。 心臟超音波 高頻率聲波聚焦 基本上,心臟震波治療是利用高頻率聲波,以反射治療聚焦於治療區塊,就好像在接受心臟超音波檢查一樣;患者躺在超音波的檢查床上,並黏上心電圖的電極片做為心律的監視。 患者的左胸壁將塗上一般超音波檢查用的人工膠,以確保震波頭與皮膚的密切接觸,震波頭治療的位置及深度由院內現有的心臟超音波的指引來進行。目前心臟震波已經通過核可引進台灣,目前亞東、高雄長庚、台北榮總皆有相關設備。 3個月9次療程 追蹤明顯改善 吳彥雯醫師指出,心臟震波整個療程將維持9個星期,其中第1、第5及第9個星期均接受震波治療,每星期接受3次的治療,視心肌缺氧範圍的大小,心臟將有3個以上不同的點接受震波治療,每點接受震波100下,每次治療需要1個鐘頭左右,無健保給付。 亞東醫院目前陸續完成3個月的療程者共有8位,4位追蹤超過6個月並完成第2次核醫心肌灌注影像的追蹤,結果顯示其中3位(75%)在治療區域缺氧範圍及嚴重度減少,與目前國內外發表的報告,約60%至70%臨床改善的結果相近。 醫療新選擇 滿意度高有禁忌 針對血管過細、過彎患者,心臟震波可改善治療困境;臨床發現,這些病人多為內科心導管手術跟外科開心手術無法或不適合治療者,甚至部分病人心臟收縮功能不佳。不過治療期間並無不適,病人可以順利完成治療,症狀及心肌掃描都有相當程度的進步,結果令人相當滿意。 【心臟震波禁忌症】: 1.心肌梗塞3個月內者。 2.病患有慢性肺疾病,影響超音波定位者。 3.心室內血栓。 4.併有急性心內膜炎,心肌炎或心包膜炎。 5.病患身上有植入裝置(心律調節器、人工瓣膜等)在震波治療位置。 6.重要全身性疾病(除心臟病)如肝硬化、末期腎臟病或惡性腫瘤等預估餘命低於6至12個月者。 7.孕婦或授乳婦。 |
Click here to edit.
|
心臟震波儀治療冠心病 國內醫界大突破 (2012-03-29)
|
|
Cardiac Shock-wave Therapy in the Treatment of Refractive Angina Pectoris
Juan Ruiz-Garcia, Amir Lerman Interv Cardiol. 2011 ;3(2):191-201. Abstract
An increasing number of patients are being diagnosed with severe coronary artery disease not amenable to traditional revascularization. Most of them remain symptomatic despite optimal medical therapy, so the resulting qualiy of life is poor. Thus, there is a need for the development of novel therapies. Shock waves (SWs) have been used in medicine for 30 years, but the recent discovery of their potential angiogenic effect has led to their most promising application in cardiovascular medicine. Cardiac SW therapy delivered to ischemic myocardium has been demonstrated to reduce angina symptoms and to improve myocardial perfusion. And although there is a lack of large series and long-term follow-up, cardiac SW therapy is by now a safe, well-tolerated procedure with no significant side effects. Introduction As a result of the improvements in both pharmacologic and revascularization therapies life expectancy for patients with coronary artery disease (CAD) has greatly increased. This means that patients with more extensive CAD are now living longer, so many of them will develop myocardial ischemia and clinical angina that are not amenable to traditional revascularization therapy.[1–4] Patients with severe, symptomatic, chronic CAD have been described as having intractable angina, end-stage CAD or have been called 'no option' patients. However, despite being considered a therapy-resistant condition, refractory angina is a more appropriate term because a considerable number of new therapeutic methods are now available. The Canadian Cardiovascular Society (CCS) has recently defined refractory angina as a persistent (more than 3 months), painful condition, characterized by chest pain caused by coronary insufficiency in the presence of CAD, which cannot be controlled by a combination of medical therapy, angioplasty and coronary bypass surgery.[5] It is necessary to ensure that revascularization is unfeasible, medical therapy is optimal and that other causes of chest pain are excluded before establishing the diagnosis of refractory angina.[5,6] Currently, few data are available on the number of nonrevascularizable patients. Estimates have been obtained from both population surveys and catheterization laboratory registries. A Swedish survey in 1994–1995 demonstrated that approximately 10% of patients referred for coronary angiography because of stable angina were rejected for revascularization despite having severe symptoms.[7] In the late 1990s, a single tertiary referral center series reported that 12% of the patients referred for symptomatic CAD were not amenable to percutaneous coronary intervention or coronary artery bypass graft.[8] A more recent study conducted in Minnesota (MN, USA) found 6.7% of patients with coronary lesions >70% and no revascularization options despite optimal medical therapy.[9] In absolute numbers it would represent more than 100,000 new patients/year in the USA and 30,000–50,000 new patients/year in Europe who will probably fulfil the diagnosis of refractory angina.[1,2,6] The outcome of these patients continues to be controversial. The controlled trials with alternative therapies conducted in patients with refractory angina demonstrated (in their control arms) that annual mortality rates varied from 3 to 21%, whereas coronary event rates (including acute coronary syndromes and hospitalization for angina) varied from 11 to 69%.[10–18] In contrast to these trials, in a large series of 1066 patients with refractory angina followed for a median of 3.5 years only 126 patients died (11.8%), and only 58 of these deaths were from cardiovascular causes (5.4%).[19] Data from 200 patients treated at our institution confirm this low rate (2% cardiac mortality per year),[20] and the recent study mentioned above also found a low mortality (15% at 3 years), although this was significantly higher than the mortality in patients completely revascularized (6.6% at 3 years).[9] Since it has emerged that the mortality of patients with refractory angina patients is less than was considered previously, it may be speculated that the primary target of the incoming therapies for this growing population should focus on relieving chest pain and improving quality of life, which is significantly impaired compared with revascularized patients,[21] and not necessarily the reduction of cardiovascular events and/or ischemia. It must also be noted that costs attributable to cardiovascular hospitalizations, outpatient visits, procedures, and medications are very high in these patients,[22,23] so new treatments are required to help to reduce the high economic burden of refractory angina. Current Treatment Possibilities in Refractory Angina It should be stated that it is of the utmost importance that all patients who are considered for these therapeutic measures are first subjected to careful optimization of medical treatment (including novel drugs with new antianginal mechanism such as ivabradine[24–26] and ranolazine[27–29]), optimum cardiac rehabilitation and evaluation concerning psychosocial pain determinants. Secondary causes of angina, such as anemia, poorly controlled hypertension, thyroid dysfunction or arrhythmias, should also be identified and treated accordingly. When evaluating new methods of treatment for refractory angina, one should consider the following points: clinical efficacy, scientific documentation, feasibility (i.e., accessibility, costs, need of compliance and follow-up) and adverse reactions. As demonstrated previously, given the likely low mortality of these patients, any novel approach should always have low complication rates. Enhanced External Counterpulsation Enhanced external counterpulsation (EECP) produces externally what an intra-aortic balloon pump does internally. Three pairs of pneumatic blood pressures cuffs are placed on the calves, lower and upper thighs. An electrocardiographic trigger is used to sequentially inflate the cuffs, starting at the calves, during onset of diastole and rapidly deflate before systole. This systematic diastolic pressure augmentation is presumed to increase coronary perfusion pressure and venous return, and to decrease ventricular afterload.[30] EECP reduces symptoms of angina and nitrate use, improves exercise tolerance and enhances quality of life of the patients with refractory angina; most of these effects are sustained for at least 3 years.[31–35] Side effects are minimal and include leg pain and bruising. Few contraindications exist, but significant aortic regurgitation, aortic aneurysm and arrhythmias that may interfere with triggering of the EECP system should be ruled our before starting therapy. The use of EECP is limited by the need for 35 days to complete the treatment.[30] Neuromodulation Spinal cord stimulation (SCS) is a recognized valid therapeutic option in Europe for the treatment of refractory angina.[6] The device has an epidural lead inserted at C7–T1 level, a subcutaneous wire and a pulse generator implanted in the left lower abdominal area. The therapy is self-administered; the patient receives stimulation for 1 h, three times daily, and can activate the device whenever angina is felt. Fewer angina attacks, reduced nitrate consumption, and improved CCS class and quality of life have been described with its use.[18,36–39] The main complications include infections (1%) and lead migration or fracture (7.8%).[39] Pain due to acute coronary syndromes has not been demonstrated to be blocked in the studies and it is probable that with SCS it continues to cause typical symptoms despite stimulation; however, special care is required with pacemakers and implantable defibrillators as SCS could interfere with some of their functions.[40–43] Cell Therapy Stem cell therapy is currently being investigated as a new therapeutic option for patients with chronic ischemia who are ineligible for revascularization. In a small randomized placebo-controlled study, myocardial injection has been demonstrated to be safe and to be associated with a modest but statistically significant improvement in myocardial perfusion, left ventricular ejection fraction (LVEF), exercise capacity and CCS class.[44] This technique is still in the experimental stages, and further studies are required to assess long-term results and efficacy for reducing morbidity and mortality. Other Therapies In contrast with EECP, SCS and stem cell therapy, disappointing results were obtained with gene therapy, percutaneous and transmyocardial laser revascularization. Opposite to their promising results in small trials, larger randomized studies demonstrated no associated benefit with their use.[11,15,45–47] One of the potential failings of these therapies may be due to their invasive approach and the potential higher risk–benefit ratio; for example, in one trial, a significantly higher 30-day myocardial infarction incidence was observed in patients receiving percutaneous myocardial laser revascularization compared with patients in the placebo group,[11] and procedure-related deaths have even been reported in some studies.[13,15] Cardiac Shock-wave Therapy
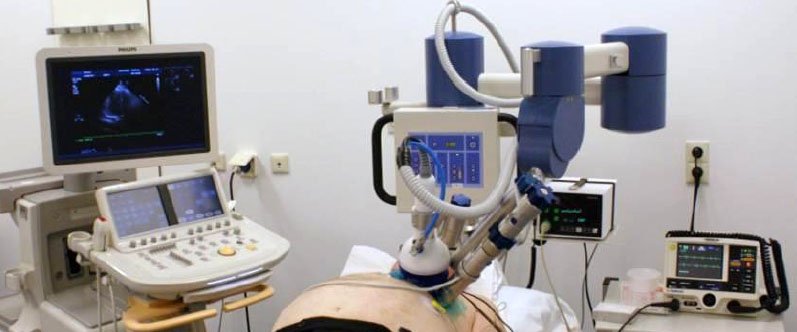
History of Shock-wave Therapy High-energy extracorporeal shock-wave therapy (ESWT) was introduced for medical use 30 years ago as a treatment for ureteral stones.[48] ESWT has changed the treatment of urolithiasis, and even today remains the primary treatment for most uncomplicated upper urinary tract calculi.[49] ESWT has also been therapeutically applied in bile ductus,[50] pancreatic[51] and salivary stones.[52] The 'destroyer-use' of high-energy shock waves (SWs) is different from the more recent 'regenerative-use' of low-energy SWs. Based on the incidental finding of iliac bone thickening in patients undergoing lithotripsy,[53] low-energy ESWT has been developed as a treatment standard or alternative therapy for a variety of orthopadic and soft tissue diseases, in several kinds of chronic tendinopathies and muscular pathologies.[54–56] The observed immediate increase in blood flow due to local vasodilation and the formation of new capillaries in the treated tissue[57–59] has led to its more promising application in cardiovascular medicine as a possible therapy for patients with refractory angina. Generation of SWs in Cardiac SW Therapy Shock waves consist of acoustic energy that can be transmitted in a liquid medium and focused with precision of several millimeters to any intended treatment area inside the body. The energy density describes the maximum amount of acoustical energy that is transmitted per pulse, and varies among different uses of SWs, from 0.09 mJ/mm2 in cardiology[60] up to 0.9–1.8 mJ/mm2 in lithotripsy.[61] Shock waves can be artificially generated by discharge of a high-voltage spark under water. Cardiac SW therapy (CSWT) is performed using a SW generator system designed to address the clinical anatomical requirements of the chest cavity. A cardiac ultrasound imaging system is used to locate the treatment area with documented ischemia. SWs are then delivered via a special applicator through the anatomical acoustic window to the treatment area under electrocardiographic R-wave gating. Several treatment sessions are required. At each session, SWs are delivered to the border of the ischemic area, to potentially induce neovascularization from the healthy area to the ischemic area. For optimal therapy, the treatment zones are divided into target spots corresponding to the size of the focal zone of the SW applicator (Figure 1). At least two CSWT generators are available today: MODULITH® SLC (Storz Medical AG, Taegerwilen, Switzerland) and Cardiospec (Medispec, Germantown, MD, USA). Figure 1. Extracorporeal cardiac shock-wave therapy. (A) The patient under electrocardiographic monitoring (B) receives, in the ischemic zones localized with ultrasound guidance, the shock waves produced by the generator that is attached to the chest wall. (C) At each session, cardiac shock-wave therapy is applied to the border of the ischemic area to potentially induce neovascularization from the healthy region to the ischemic area. Cardiac SW Therapy Results Nishida et al. published the first study to examine the effects of CSWT that could induce angiogenesis in the heart.[60] Initially, they treated human umbilical vein endothelial cells with SW at different energy levels, and found that CSWT significantly upregulated mRNA expression of VEGF and its receptor Flt-1, with a maximum effect noted at 0.09 mJ/mm2. Subsequently, they performed an experiment in a porcine model of chronic myocardial ischemia without causing myocardial infarction. On the basis of the in vitro experiment, they applied a low-energy SW (0.09 mJ/mm2) to nine spots in the ischemic region (200 shots/spot). Patients treated with CSWT three-times per week were compared with the control group, who received the same anesthesia procedures three times a week but without the CSWT. A total of 4 weeks after the CSWT, patients who received SW therapy, but not the control group, had a marked development of coronary collateral vessels in the ischemic region and a significantly increased number of visible coronary arteries in the region and this was associated with a complete and significant recovery of the ejection fraction only in the SW group. The CSWT also significantly normalized the regional myocardial blood flow, increased capillary density and upregulated VEGF expression in the ischemic myocardium in vivo (Figure 2). Owing to the promising results in animal studies, Fukumoto et al. started a small, non-placebo-controlled clinical trial of CSWT in nine patients with refractory angina.[62] A CSWT consisting of 200 shots/spot at 0.09 mJ/mm2 for 20–40 spots each time, three times a week/series, was applied over the ischemic area, confirmed by dipyridamole stress thallium scintigraphy, and repeated up to three series at 0 and 1, 3 or 6 months depending on the results of their evaluation. A significant relief of the symptoms, as evaluated by CCS class score and the use of nitroglycerin (NTG), was confirmed after the CSWT. In addition, a clear improvement in myocardial perfusion, evaluated by dipyridamole stress thallium scintigraphy, was observed in the ischemic myocardium, but only where SW were applied. It should be noted that the anti-ischemic effects of CSWT started at 3 months after the therapy and persisted for at least 12 months (end of the follow-up period). Figure 2. Shock-wave effects. SW treatment increased the mRNA expression of VEGF and its receptor Flt-1 in human umbilical vein endothelial cells, with a maximum effect at 0.09 mJ/mm2. (A) Normalization of the LVEF was only observed in the group of pigs treated with the SW therapy (B). The regional myocardial blood flow evaluated with colored microspheres in both the endocardium and the epicardium also improved significantly in the SW group (C). LVEF: Left ventricular ejection fraction; RMBF: Regional myocardial blood flow; SW: Shock wave. Reproduced with permission from [60]. Khattab et al. in Germany shared their initial experience with CSWT applied in ten patients with refractory angina 1 year later.[63] All ten patients had evidence of myocardial ischemia on exercise Tc99 SPECT perfusion scan. They were subjected to nine sessions (three cycles, with the same energy and amount of shocks described previously[60]) over 3 months. A significant reduction in symptoms (CCS class at baseline was 3.3 ± 0.5 and at follow-up was 1.0 ± 1.3) and myocardial perfusion improvement was reported. Myocardial perfusion improved again only in the ischemic areas treated with CSWT. Recently, the same group from Japan has published a small, unique double-blind placebo-controlled trial in eight patients with refractory angina.[64] Patients were treated with one series of placebo and CSWT in a double-bind and crossover manner with an interval of 3 months. One series of CSWT (200 shoots/spot at 0.09 mJ/mm2 for 40–60 spots per session) comprised three sessions per week. Follow-up was limited to 3 months. CSWT, but not placebo, significantly improved symptoms, NTG use and 6-min walking distance. Furthermore, the LVEF and left ventricular stroke volume, evaluated by MRI, improved significantly only after CSWT. Currently, to the best of our knowledge, there are few other published data about the application of CSWT in patients with refractory angina. There are some isolated cases reported in journals[65] and small cohorts of patients presented at congresses.[66–73] It should be mentioned that in all of these studies, a positive effect of CSWT in clinical symptoms was shown and an improvement in myocardial perfusion – assessed by PET or SPECT – was demonstrated. These clinical results are supported by animal experiments conducted in pigs in a model of acute phase of acute myocardial infarction (AMI). A significant recovery of LVEF and reduction in left ventricular end-diastolic volume was described at 4 weeks after the CSWT in the SW group compared with the control group. In addition, regional myocardial blood flow and the number of capillaries in the border zone of the infarction were significantly improved in the SW group. These beneficial effects were only observed when the CSWT was started 3 days after AMI, but not if started 4 weeks later.[74] Similar results were recently obtained in pigs subjected to 90-min ischemia and reperfusion.[75] In a different rodent model, direct epicardial SWs applied 4 weeks after AMI induced significant angiogenesis and improved ventricular function compared with the control group.[76] Taking into account these previous experimental studies, Vasyuk et al. have recently conducted a first study in patients with ischemic heart failure due to documented myocardial infarction (at least 6 months before inclusion).[77] A total of 24 stable patients with systolic dysfunction (LVEF <40%) and no planned revascularization received CSWT (nine sessions with 100 shocks per spot per session at 0.09 mJ/mm2 energy level in hibernated or ischemic segments detected by dobutamine stress echocardiography) and were followed for 6 months. CSWT significantly decreased New York Heart Association class, CCS class and NTG use per week. Similarly, an improvement in 6-min walk test and quality of life was reported. The authors also found a significant increase in LVEF and in perfusion scores after the CSWT. The available human data are summarized in Table 1. Possible Mechanisms of Action Although the precise mechanisms remain to be elucidated, two major effects may contribute to the aforementioned observations: immediate vasodilatation, and the induction of neovascularization in the treated tissue, which probably accounts for the observed long-term effects. It has been described that SW may induce tissue cavitation,[78] generating highly localized physical forces that could produce localized stress on cell membranes. This would lead to a variety of biochemical effects, including: shear stress on cell membranes,[79] hyperpolarization and Ras activation,[80] an increase in nitric oxide synthesis,[75,81–83] an upregulation of VEGF, its receptor Flt-1 and PGF,[60,76,84–86] in addition to an enhance expression of stromal-derived factor-1.[87] Another potential cellular mechanism may involve the recruitment of progenitor cells to the site of the ischemia undergoing CSWT.[87–89] Thus, we can conclude that there are probably multiples angiogenic pathways involved in the beneficial effects of CSWT (Figure 3). Figure 3. Proposed mechanism for the beneficial effects of cardiac shock-wave therapy. The observed clinical effects are believed to be related to different angiogenic pathways, including NO, VEGF, PGF, (SDF-1) and recruitment of EPCs. EPC: Endothelial progenitor cell; NO: Nitric oxide; SDF-1: Stromal-derived factor-1. Adverse Effects No significant side effects or symptoms were reported during the CSWT.[62,64–67,69] Some of the patients referred experienced transient dizziness and warm sensation[71,73] and in one case the procedure was not completed due to the painful sensation.[63] Only in one case was a slight troponin T elevation found;[63] in the rest of the cases, elevation of myocardial enzymes in animal and human studies was not observed.[60,65,66,68–70,73,77] Currently, there is no available information about human histology, but in rats no histologic signs of myocardial damage, including cellular infiltrates, extravasates, edema, cell membrane, or cell nuclei damage and hypertrophy, were observed.[76] In human cardiac MR studies, no increase in scar tissue volume was demonstrated following CSWT in humans.[71,73] The CSWT therapy was delivered under electrocardiographic R-wave gating, and no significant arrhythmias were reported during it. However, attention should be paid in patients with pacemakers or implantable defibrillators, as there are not sufficient safety data. Advantages A major advantage of CSWT over other angiogenic strategies (cell therapy, gene therapy and laser revascularization) is the fact that it is non-invasive and can be performed as an out-patient treatment. It can be repeated easily as no surgery, anesthesia or catheter intervention is required. As described previously, the prognosis of these patients seems to be better than previously thought, so it is important to highlight that no major adverse effects have been reported with its use. In the absence of long-term follow-up, it suggests an important positive benefit/risk balance of this approach. Limitations As treatment area needs are localized, the patients without an adequate echocardiographic window (e.g., pulmonary disease, breast prothesis or overweight) cannot receive this therapy, and the safe use of CSWT in patients with pacemakers or implantable defibrillators is still not defined, but beyond these points no other technical limitations are described. However, as the number of sessions to observe the benefits is high and variable, some concern has been raised regarding the adherence of patients to CSWT, similar to EECP therapy. It is important to note that CSWT is still an experimental therapy, with very few patients currently being treated with this modality around the world, compared with other strategies used in refractory angina, and the long-term prognosis still remains unknown. Besides, as there is a lack of large placebo-controlled studies, it might be possible that the short-term reduction in symptoms observed were mediated by a placebo effect. Thus, there is a requirement to perform multicentric, randomized and/or placebo-controlled studies, with long-term follow-ups. Conclusion Cardiac SW therapy is a novel approach that might play an important role in the treatment of patients with refractory angina. It seems to reduce the symptoms and to improve the quality of life. As it is a noninvasive therapy and no significant adverse effects have been reported with its use, it emerges as an attractive option for these patients. However, there is still little experience with the use of this technique compared with EECP and SCS, so more data deriving from randomized and placebo-controlled trials are required before its widespread use can be implemented. |